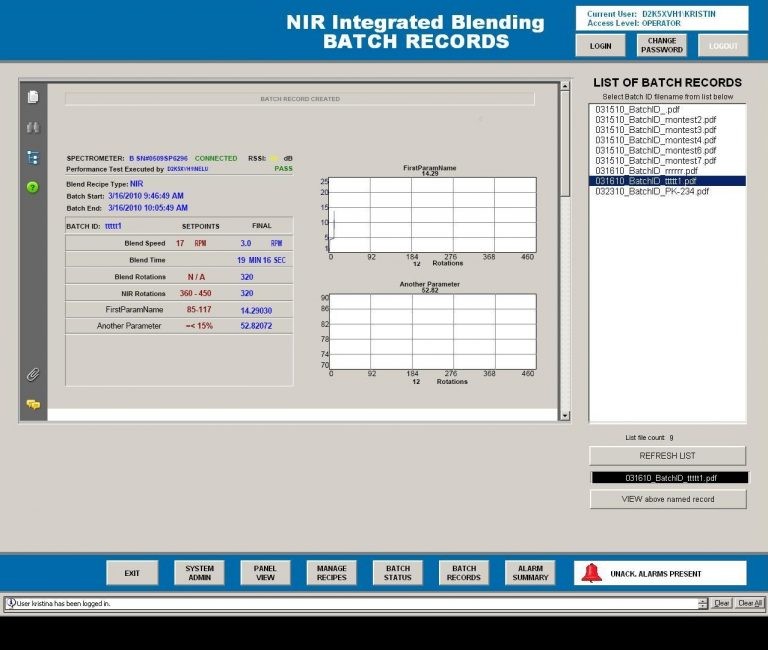
A major pharma company had a corporate directive to implement PAT for the purpose of real time API release – The CPS solution provided three significant benefits.
The previous system would stop blending after a specified number of rotations and then the product would be set aside in inventory until the QC lab could conduct solvent based tests confirming the product quality. Out of spec product was returned to the blender for additional blending and then retested. The PAT solution benefits were 1) reduced capital/inventory by reducing inventory while waiting QC test results, 2) reduction of product testing costs by reducing labor and solvent hazardous waste disposal and 3) shorter production times.
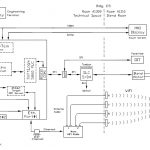
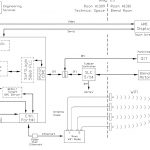
The CPS PAT process solution used standard off the shelf software products that allowed the client to make adjustments to the chemometric and end point determination calculations. The use of OPC communications drivers allows any test instrument to be used in the system.
- Wireless Connection of the NIR sensor (rotates with the blender)
- Third Party Off-The-Shelf Software products instead of traditional proprietary software
- Able to work with any OPC compliant instrument (FTIR, NIR, Chromatograph, Spectrometer, etc.)
- Real Time Release of API to Manufacturing